Rocky Vista University, College of Osteopathic Medicine, Ivins, Utah, USA.
Correspondence: brianna.landis@rvu.edu
Conflict of interest statement: The author declares no potential conflicts of interest.
Keywords: primary lens tumor; tumorigenesis; cancer prevention
Despite the mitotically active nature of the ocular lens and near-constant exposure to ultraviolet radiation, there have been no reported cases of primary tumors in the human lens. In contrast, such tumors have been induced and reported in the lens of non-human vertebrates, particularly in cats. This report discusses various theories, including the avascular nature of the lens, the presence of barrier properties within the ocular environment, and the lens capsule composition as a potential chemo-mechanical barrier against tumorigenesis. Despite the significant implications for cancer prevention and treatment, there has been limited research into this phenomenon. Identifying protective mechanisms could contribute to a better understanding of human cancer genetics and potentially lead to preventative treatments.
There are no reported cases of primary tumors within the human lens, a phenomenon that is poorly understood and scarcely investigated. This perspective highlights theories for potential tumor-resistant properties and urges researchers to continue investigating. Further research could have significant implications for understanding cancer biology, tumorigenesis, and future preventative treatments.
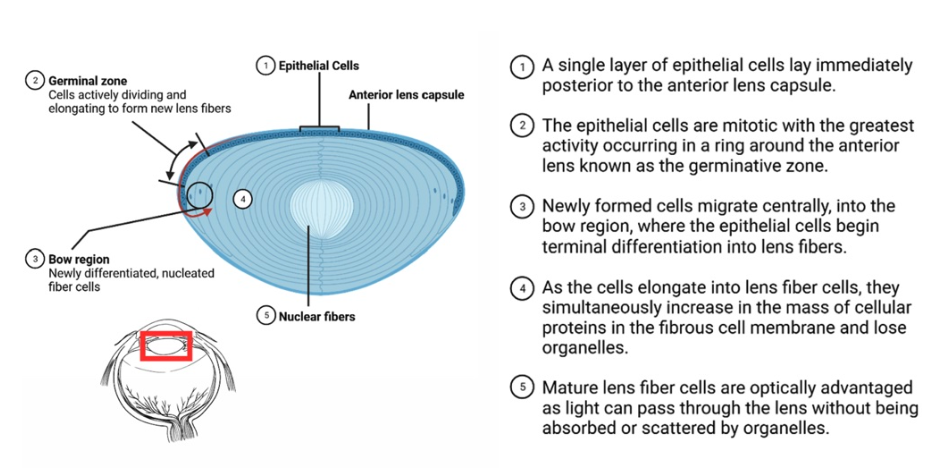
Over the past century, the absence of primary tumors of the human lens has been remarked upon in the literature but never rigorously investigated (1–4). If true, this observation is noteworthy and certainly remarkable because all dividing cells, even in invertebrates, can develop genetic mutations and form tumors (5). Immediately posterior to the anterior lens capsule lies a single layer of epithelial cells. Within this layer is the germinative zone. As cells divide, they migrate laterally and then displace centrally, losing organelles as they do so, so that they can laminate and incorporate into the clear crystalline lens that refracts light onto our retina (Figure 1) (6). By design, this mitosis occurs throughout life and into old age, thus it is curious that such a mitotically active tissue, with near constant exposure to ultraviolet radiation, is seemingly resistant to tumorigenesis.

Limited research has explored this phenomenon, the results of which are equally perplexing. Malignant tumors of the lens can be induced with exposure to carcinogens (7) and oncogenic viruses (8) and can be engineered genetic defects in transgenic animals (9–11). Strikingly, malignant tumors of the lens can occur spontaneously in other nonhuman vertebrate species (cats, rabbits, dogs, and birds) (12–20).
However, no case of malignant or benign tumor of the human lens has been reported or described in the literature. A review of veterinary databases revealed that in non-human species, malignant tumors of the lens occur most commonly in cats, constituting 4.5% of intraocular and adnexal neoplasms in that species (20). It is established that rupture of the lens capsule is a major risk factor for the development of the tumor, coining the nomenclature of feline ocular post-traumatic sarcoma (a benign neoplasm of lens epithelial origin) (14). Retrospective review of previously unreported cases of primary lens tumors in cats from the University of Wisconsin School of Veterinary Medicine's Comparative Ocular Laboratory collection reveals that all cases showed evidence of lens capsule rupture and most had some degree of uveitis, similar to tumors observed in other vertebrate species (birds, rabbit, and dog) (20).
With awareness of the strong correlation of capsular trauma to tumorigenesis, it is even more shocking that such tumors have not been described in humans. Cataract removal is the most commonly performed surgical procedure in humans (21), and by design, the lens epithelium is traumatized and retained during modern extracapsular cataract extraction. Despite frequent surgical injury to the lens epithelium, no benign or malignant lens tumors have been documented (20). Could genetic protective mechanisms exist? And if so, wouldn't identifying this mechanism hold significant implications to better understanding the genetics of human cancers and ultimately providing preventative treatment?
Several theories have arisen to attempt to explain this phenomenon. The lens is a naturally avascular tissue, sensibly designed to minimize the scattering of light as it passes through to the retina. As such, the lens acquires nutrients from aqueous and vitreous components passing through the semipermeable membrane of the lens capsule (22). It has long been proposed and accepted that adequate vascular supply is essential for tumorigenesis and progression (23). Solid tumors, irrespective of their source, typically begin as a small cluster of cells relying on nutrients diffusing from nearby tissues (23). As the tumor grows, it eventually reaches a size where simple diffusion is inadequate to support further growth and angiogenesis is needed to facilitate further growth. Hence, it is sensible to assume an avascular tissue, such as the lens, could harbor potential tumors to a minimal size dependent on available resources. Interestingly, however, even proangiogenic colonies of neoplastic cells have not been described (24).
Additionally, another avascular ocular tissue, the cornea, can still be invaded by advancing tumors maintaining their angiogenic factors (25). As such, avascularity alone does not entirely explain the lack of primary tumor formation in the lens. However, unlike the lens, the cornea lacks a capsular barrier. Although, it is hypothesized that the Bowman's layer of the cornea serves as a form of corneal barrier, as tumors in the stroma layer beneath are largely undiscovered despite a notable prevalence of chromosomal abnormalities (26). Hence, it is conceivable that either the ocular environment or the existence of barrier properties could contribute to the absence of tumor development in the lens.
Other theories emphasize this possibility by suggesting the lens capsule is a chemo-mechanical barrier. Among other molecules, the lens capsule is largely composed of collagen types I-IV (27, 28). Fragments of collagens make up endostatins, which are known to act as inhibitors of angiogenesis (29). It is postulated that these endostatin molecules exist near or within the lens capsule to serve a protective mechanism against angiogenesis, both for the purpose of preserving lens transparency but also inhibiting the angiogenesis of tumors (29). Notably, there is evidence indicating that fragments of type IV collagen, the primary constituent of the lens capsule, may impede tumor cell growth (30) and hinder the activation of matrix metalloproteinases (31) in tumor cells believed to contribute to invasiveness. This evidence lends support to the idea that a growth inhibitor associated with the lens capsule could prevent neoplastic transformation in subcapsular epithelial cells.
Interestingly, even highly invasive melanomas and retinoblastomas, sometimes occupying the entirety of the posterior chamber, demonstrate well-defined borders at the lens capsule interface (24). These types of tumors are widely recognized for their infrequent invasion or direct contact with the lens capsule. Instead, the tumor-lens interface becomes filled with debris and fluid (24). Is it possible that these tumors are repelled by an unknown chemo-mechanical property of the lens capsule?
Undeniably, these observations are quite remarkable and impress exciting potential for research to advance cancer prevention and treatment. So then why is there such limited research into this phenomenon? According to NIH.gov, in 2020, the National Institute of Health (NIH), a major research funding agency in the United States, estimated a cost of nearly 6.5 billion dollars to support cancer research efforts. However, a search into the NIH RePORTER database revealed that the NIH has never funded any projects attempting to investigate the seemingly tumor-resistant properties of the human lens capsule epithelium (32). Such research seems to be a prime candidate for identifying a cancer-inhibiting gene or genes in humans. With current genetic techniques, it should be possible to identify the genes involved in lens tumor formation in other species and to use these as candidate genes in identifying the genes responsible for preventing cancer in the human lens. A genetic protective mechanism is hypothesized to exist, and identifying this mechanism may be of significant value in enhancing the understanding of human cancer genetics.
Given the striking absence of primary tumors in the human lens, several experimental approaches could be employed to elucidate the underlying protective mechanisms, such as comparative genomic transcriptomic analysis, CRISPR-based functional genomics, lens capsule extracellular matrix components, in vivo animal models, epigenetic and regulatory network studies, and organoid and 3D cell culture models (Table 1).
Table 1 | Potential experimental methodologies and genetic approaches for investigating protective mechanisms of the human lens and their associated research benefits.
| Experimental approach | Benefits of the proposed method |
|---|---|
| Comparative genomic transcriptomic analysis | Whole-genome and transcriptome sequencing of human lens epithelial cells compared with lens epithelial cells from species known to develop lens tumors (e.g., cats) could reveal key genetic differences. Identifying differentially expressed genes, particularly those involved in tumor suppression, DNA repair, and apoptosis, could pinpoint genetic factors responsible for the lens' tumor-resistant properties. Additionally, single-cell RNA sequencing (scRNA-seq) could provide insights into unique gene expression profiles of subpopulations within the lens epithelium. |
| CRISPR-based functional genomics | CRISPR-Cas9 or CRISPR interference (CRISPRi) could be used to selectively knock out or suppress candidate tumor suppressor genes in human lens epithelial cell cultures to determine their role in preventing neoplastic transformation. Conversely, overexpressing these genes in other epithelial cell types prone to tumorigenesis could help assess their protective effects beyond the ocular environment. |
| Lens capsule extracellular matrix (ECM) analysis | The lens capsule's composition, particularly its type IV collagen content and potential angiogenesis inhibitors like endostatins, could be further explored using proteomics and mass spectrometry. In vitro studies exposing cancer cell lines to isolated lens capsule components could assess whether these elements exert direct tumor-suppressive effects. Additionally, matrix metalloproteinase (MMP) activity assays could determine whether the capsule actively prevents ECM degradation, a key step in tumor invasion. |
| In vivo animal models | Transgenic animal models could be developed to express human lens-specific genes in species that normally develop lens tumors. If these genes confer tumor resistance, it would provide strong evidence for their protective role. Additionally, lens capsule transplant experiments in animal models with aggressive ocular tumors could test whether the human lens capsule creates a hostile environment for tumor growth. |
| Epigenetic and regulatory network studies | Investigating DNA methylation patterns and histone modifications in human lens epithelial cells could reveal whether epigenetic regulation contributes to their resistance to tumorigenesis. Chromatin immunoprecipitation sequencing (ChIP-seq) could help identify key transcription factors involved in maintaining the lens' quiescent but mitotically active state without leading to malignant transformation. |
| Organoid and 3D cell culture models | Engineering lens epithelial organoids or 3D co-culture systems with cancerous cell lines could provide a controlled environment to study potential tumor-inhibitory effects of the lens microenvironment. This approach could be particularly useful for testing the effects of biochemical signals and mechanical properties of the lens capsule on tumor suppression. |
By employing these methodologies, researchers could move beyond theoretical explanations and begin identifying actionable molecular targets for preventing tumorigenesis in other tissues. Understanding how the lens naturally resists tumor formation could lead to the development of novel cancer therapies, including biomimetic extracellular matrices, anti-angiogenic compounds, and gene therapies designed to enhance tumor suppression in high-risk tissues.
The apparent resistance of the human lens epithelium to tumorigenesis presents an untapped avenue for oncological research, with potential implications for cancer prevention and treatment. The absence of primary tumors in a tissue that remains mitotically active throughout life, despite continuous exposure to ultraviolet radiation and surgical trauma, suggests the existence of unique protective mechanisms. If genetic or biochemical factors within the lens epithelium or capsule contribute to this resistance, identifying these mechanisms could inform strategies for suppressing tumorigenesis in other tissues. For example, if the lens capsule's extracellular matrix components, such as type IV collagen and endostatins, play a role in inhibiting angiogenesis and tumor invasion, similar mechanisms might be leveraged therapeutically in cancers reliant on angiogenic signaling. Moreover, uncovering genetic factors that prevent neoplastic transformation in the lens could contribute to the identification of novel tumor suppressor genes, expanding our understanding of intrinsic cancer resistance in humans. This phenomenon underscores the necessity of further research, as elucidating these protective mechanisms could inspire innovative approaches to cancer prevention and treatment, shifting the focus from reactive therapies to proactive, biologically informed interventions.
The author would like to acknowledge Anthony Pappas, Ph.D. for introducing this fascinating lapse in knowledge, as well as student doctors Parker Webber and Bosten Loveless for their contributions to the poster presentation Paradoxical lack of investigation into the natural tumor-resistant properties of the human lens capsular epithelium. Their efforts were paramount in sparking interest in this subject and highlighting this notable gap in the literature.