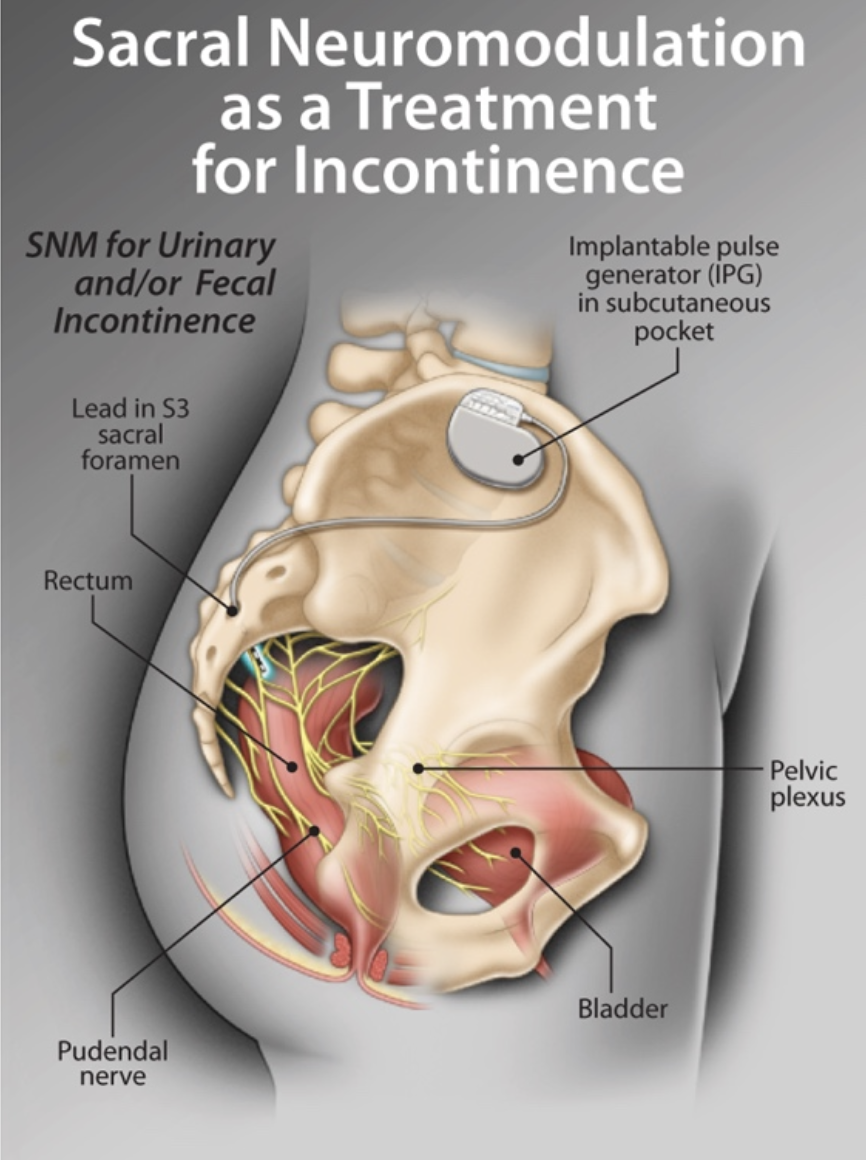
Sacral Neuromodulation (SNM) is an established nerve stimulation treatment for Functional Gastrointestinal Disorders (FGIDs) such as Faecal Incontinence (FI), Slow Transit Constipation (STC) and ileus. SNM may involve the surgical placement of electrodes against the sacral roots of the spinal cord to alter the physiological function of the target organs. It can also be performed by using Transcutaneous Electrical Nerve Stimulation (TENS) which is a non-invasive and cost-effective SNM technique consisting of placing electrodes on the intact skin instead of implantation. However, despite the increasing application of this therapy, the neural underpinnings of SNM are poorly understood. One of the consequences of this poor understanding is that currently SNM is administrated on a trial-and-error basis and there are no standard guidelines on the optimal therapeutic parameters. Evolving studies aim at exploring the neural mechanisms of SNM for FGIDs by either focusing on the extrinsic innervation of the gut or probing the relationship between the gut and the central nervous system (CNS). Recent studies have revealed that various brain regions could be involved in the process. Moreover, detecting meaningful electrical changes over the somatosensory cortex of SNM treated patients indicates that C-afferent fibers might be the most likely target nerves of SNM. Nevertheless, a deeper understanding of the underlying neural function of SNM is essential to improve identification of the most effective stimulation techniques and parameters for FGIDs which can subsequently influence the decision of clinicians for permanent implantation of electrodes.
Sacral Neuromodulation (SNM) was first used clinically in the early 1980s to treat patients with urinary incontinence and retention (9). SNM involves the surgical placement of electrodes against sacral roots of the spinal cord to modulate the physiological function of the target organs through nerve stimulation (Fig.1). It can also be performed using Transcutaneous Electrical Nerve Stimulation (TENS) which is a non-invasive and cost-effective neuromodulation technique consisting of placement of electrodes on the intact surface of the skin. TENS is an ideal alternative to electrode implantation as similar results can be achieved while avoiding infection and other complications associated with the surgical procedure (16).
TENS therapy was initially introduced to manage painful conditions. Hence, it was not extensively used to treat non-painful conditions such as functional gastrointestinal disorders (FGIDs). In 1995 Matzel & colleagues used SNM for the first time to treat Faecal Incontinence (FI) (11,15). Since then, SNM has been evaluated and used by clinicians to manage FI, chronic constipation and ileus. It has specially become a first line therapy for FI with a success rate of 74-86% (4,11,15).
Nevertheless, despite the increasing application of this treatment, the neural underpinnings of SNM are poorly understood specially with respect to FGIDs (9,11,15). The main paradox about SNM's physiological function is that not only it can treat a wide range of disorders, but it is beneficial for disorders with divergent symptoms. It is intuitively difficult to fathom how SNM can benefit both FI and chronic constipation which are disorders with contrasting pathophysiology.
An increasing number of studies have explored SNM's underlying mechanism of action in both clinical settings and animal models (8,9,11,12). Some postulate that SNM modulates the balance between afferent and efferent nerve tone whereas others have demonstrated a central cortical effect (10,11). However, most data on the neural function of SNM are derived from the field of urology (10,15). The activation of afferent inhibitory pathway is generally accepted as the underlying neural mechanism of SNM in urinary incontinence and idiopathic detrusor instability. Studies have also revealed that stimulation of afferent fibres in the pudendal nerves inhibits the overactive bladder (17,22).
Although research on the outcomes of SNM for urinary tract ailments can provide insights into the workings of SNM for FGIDs, it is not enough to identify its neural mechanisms for such disorders. Over the past two decades, several hypotheses have been advanced to explain the beneficial effects of SNM for FGIDs. These can be classified into two main research focus areas of extrinsic innervation of the gut and the central nervous system.

There is much evidence that somato-visceral reflexes mediate colonic activity (5,10, 25). Therefore, several studies on the effect of SNM on both FI and chronic constipation have relied on this hypothesis (5,10,24,25). For instance, a colorectal electromyographic activity experiment by Vitton et al., (2008) demonstrated that stimulation of sacral dorsal roots in cats inhibits the colonic spike potential frequency. This effect faded away by administrating a noradrenergic blocker and sectioning the sympathetic efferent fibres indicating that the sympathetic system was involved in the process (25).
Clinical studies on FI patients have provided support for this hypothesis; though the existence of a somato-visceral reflex in humans is not as clear as in animals (10). Studies by Michelsen et al., (2010) on the postprandial effects of SNM in FI patients revealed a significant increase in retrograde transport in the descending colon and a modest increase in total colonic transit time in patients who received SNM therapy. This implies the existence of a sympathetic inhibition reflex that is activated to improve continence (20).
However, other studies on FI patients have reported varying results. For instance, in a study by Uludag et al., (2006) anal resting and squeeze pressure as well as segmental and total colonic transit time in FI patients did not show any significant difference after SNM. Moreover, studies on patients with slow-transit constipation provide evidence for a contrasting neural mechanism (24). Dinning et al., (2006) reported that SNM can induce colonic propulsive activity which is triggered by parasympathetic system rather than sympathetic in constipated patients. Additionally, a study on mucosal blood flow revealed that SNM can increase blood flow to the colonic mucosa which is achieved by enhancing parasympathetic activity (10).
A possible explanation for such discrepancies is that SNM modulates both sympathetic and parasympathetic systems and the observed effects are the net result of this dual activation. In addition to somatic afferent fibres, parasympathetic efferent fibres could also be activated to achieve the balance between afferent and efferent nerve tone. Somato-visceral dual activation hypothesis has been convincingly evaluated for urinary tract disorders (9,23). However, future research is required to determine if SNM for FGIDs results in this dual activation.
This hypothesis suggests that SNM inhibits the activity of C-afferent fibres that form the ascending limb of the defecation reflex. SNM blocks the transfer of sensory signals to the brain stem and results in reduced defecation. Anatomically, the fibres originating from S2-S4 spinal segments comprise of the pelvic nerves, the pudendal nerve (somatic fibres) and afferent sensory fibres innervating the colon, rectum and anal sphincters. Therefore, SNM may potentially stimulate both somatic and autonomic efferent and afferent fibres. However, there is much evidence that the afferent sensory fibres are the most likely neural targets of SNM (8,10,11,21). This hypothesis is strongly supported by data that detected the cerebral effects of SNM over the somatosensory cortex (8,9,10,11,15).
Additional supporting evidence for this hypothesis comes from decades of TENS studies on painful conditions demonstrating that neuromodulation blocks incoming afferent information in peripheral nerves. Impulses generated in afferents by TENS travel towards the sensory receptors and collide with impulses generated during activation of sensory receptor cells by natural stimuli that are travelling to the CNS. Therefore, afferent information is extinguished during TENS therapy (16). Moreover, high intensity TENS activates small-diameter myelinated afferents that have higher thresholds of activation. TENS-induced activity in these afferents leads to excitation of inhibitory interneurons preventing the transmission of nociceptive information to the CNS (16).
While this hypothesis elucidates how SNM might aid FI patients, it provides little explanation for the beneficial effects of SNM for constipation. If SNM blocks the C-fibres in the ascending limb of the defecation reflex, then how are constipation symptoms improved through SNM therapy where colonic propulsive motility is enhanced? This question requires further investigation.
Modification of the activity of external anal sphincter (EAS) could be one of the reasons that SNM boosts faecal continence. This hypothesis maintains that SNM activates somatic large afferent fibres that enhance EAS activity leading to improved continence. Nevertheless, it was magnetic stimulation studies that gave rise to this hypothesis not SNM.
Harris et al., (2008) demonstrated that chronic lumbosacral magnetic stimulation increased the amplitude of cortico-anal electromyographic response which implies that a supraspinal mode of action is responsible for increasing EAS squeeze pressure in FI patients. However, results from this study could not be replicated in patients who were treated by SNM. Additionally, anorectal manometry failed to identify any significant change in the EAS activity after chronic SNM therapy in FI patients. Yet, several studies recording cortical activities, detected the activation of motor cortex during SNM administration which is consistent with this hypothesis (10,13).
Apart from lack of substantial evidence that SNM modulates EAS activity, this hypothesis is not enough to explain how SNM works for such a wide range of FGIDs. If more supporting data is gathered, this hypothesis can at best help in understanding how SNM enhances EAS activity in FI.
Recently with the advancement of brain imaging techniques, a clear effect of SNM on the CNS has been detected (1,9-11,18). Although research on central effects of SNM for FGIDs is deficient, there is strong electrophysiological evidence from TENS studies on both painful and other non-painful conditions for the central effects of SNM. Evolving animal and human data reveal specific changes in cortical activation following SNM treatment specially over the somatosensory cortex using Electroencephalogram (EEG), PET scan or MRI (8).
For instance, a series of pain studies by Garisson & Foreman (1994, 1996, 2002) on anesthetized cats using microelectrodes to record extracellular action potentials found that TENS reduced activity in 65% of spontaneously active spinal dorsal horn cells. These results were reproduced in several other animal studies (16). They later postulated that the mechanism of action of TENS includes inducing activity in small-diameter cutaneous and muscle afferents. This activity excites central transmission neurons in ascending neuronal tracts which synapse in regions of the brain that are part of the descending pain-inhibitory pathways and form a feedback loop to the spinal cord to prevent further transmission of noxious information (16).
One of the most comprehensive studies on the cerebral effects of SNM for FGIDs was performed by Giani et al., (2011) who evaluated the cortical effects of SNM (percutaneous insertion of a temporary electrode into the sacral nerve root at S3) after pudendal nerve stimulation administrated to both FI and constipated patients. Electrical cortical responses were recorded before and after chronic SNM therapy using a mere two electrode EEG. Evaluation of the latency of the somatosensory evoked potentials (SEP) induced by pudendal nerve stimulation took place before and one month after SNM therapy (9). Results demonstrated a significant decrease in the latency before the first positive electric deflection (P40) over the somatosensory cortex in patients of both groups who were successfully treated with SNM as well as in the first negative deflection (N50) in FI patients (9,15). These results indicate that SNM mainly targets afferent sensory fibres. Moreover, it implies that measurement of P40 and/or N50 SEPs using a non-invasive EEG test may help to predict the outcome of SNM which subsequently influences the decision of clinicians for permanent implantation of electrodes.
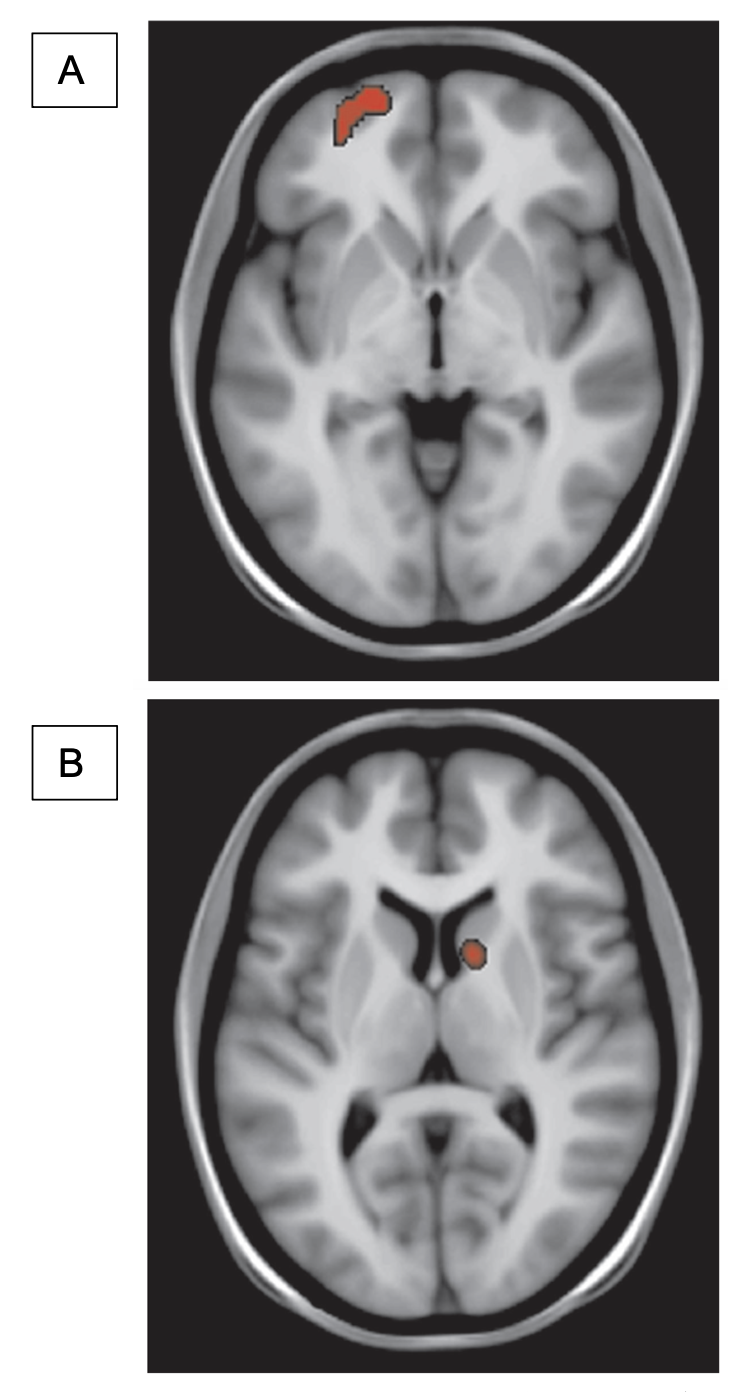
Using PET scan and MRI, Lundby et al., (2011) inferred that the afferent projections of colorectum are the target of SNM in FI patients. They measured local increase of regional cerebral blood flow (rCBF) caused by increased neural activity through analyzing changes in the uptake of a radiolabeled tracer of blood flow. The initial stimulation of sacral nerves through an implant activated a region on the contralateral frontal cortex but after chronic stimulation this activation was shifted to ipsilateral caudate nucleus which is a region of the brain involved in learning and reward processing (Fig.2). A similar cerebral effect has also been observed in vagus nerve stimulation (18).
To establish optimal stimulation parameters, Evers et al., (2014) conducted an animal experiment in which anal canal SEPs were recorded using a flexible multi-electrode array inserted in the brain of anesthetized rats. For SNM stimulation, a needle electrode was placed in the first sacral foramen and a wide frequency range of 0.1 to 100 Hz and various durations were applied. Results revealed that the magnitude of evoked potentials over the somatosensory cortex depended significantly on SNM stimulation frequency. Even a short burst could result in a long-term potentiation in the somatosensory cortex. However, to produce this effect, a critical burst duration of at least 3 minutes was required (8).
The effect of stimulation frequency and amplitude on SNM's outcome has also been studied in humans. Duelund et al. (2013), examined different pulse combinations for 4 weeks on FI patients and more than half of the patients preferred higher frequency stimulation of 31 Hz to increase total number of spontaneous bowel movements per week (6).
Remarkably, various clinical TENS studies on constipation, FI and ileus suggest a wide range of optimal stimulation parameters for the same disorder (6,16). As pointed out by Devane et al., (2015), the optimal stimulation parameters are so highly variable in the literature that it is impossible to establish any standard guidelines. On the other hand, in most human studies higher frequencies and longer durations provided better results while in animal studies the trend is towards lower frequencies (8). This casts doubt on the transferability of results from animal models to humans. However, different methodological issues may be partly responsible for this observed discrepancy. For example, in most animal studies only a brief time period is examined in contrast to the chronic nature of SNM therapy in humans (8,10).
Another line of research on central effects of SNM is focused on the expression of a protein product of proto-oncogene c-Fos which is expressed in response to direct stimulation by neurotransmitters and has been long used to label activated neurons in the CNS. It is known that electrical stimulation of the pelvic nerves induces c-Fos expression in dorsal horn neurons of the spinal cord (14). Ishigooka et al., (2002) investigated the effect of SNM on c-Fos expression within the spinal cord in rats with or without lower urinary tract irritation who underwent SNM and c-Fos immunohistochemistry of the spinal segments. Results revealed that the animals treated by SNM had a significantly higher c-Fos positive cells in L6 and S1 spinal segments (14). However, to my knowledge, no such studies have been conducted on SNM for FGIDs.
Despite the increasing body of research, there is not yet sufficient information neither about the central effects of SNM on the clinical outcome for FGIDs nor the most effective parameters to be administrated. One of the consequences of this poor understanding is that currently SNM is applied to the GI patients on a trial-and-error basis. The excruciating process may involve going through a test period of 2-4 weeks (or longer) of surgical temporary electrode implantation to evaluate the potential benefits of SNM therapy before implanting permanent electrodes (8,10,11).
Oddly enough, the optimal stimulation parameters used for urinary tract disorders have been directly transferred to gastroenterology without adaptation which indicates lack of sufficient research in the field (8). In addition, the gap in knowledge about underlying neural mechanisms of SNM has led to the uncertainty of whether sensory or motor results are the best predictor for treatment success (11,15). Although most clinical studies have been focused on the cortical representation of the anal sphincter pathway and somatosensory cortex excitability, animal studies suggest the involvement of multiple brain structures such as dorsal brainstem and caudate nucleus (15,18). Such observations are more consistent with results obtained from studies on patients with urinary incontinence (1,9,11).
More importantly, results of the effect of SNM on somatosensory cortex seem conflicting (11,15, 22). While some studies show increased cortical excitability (13), others reported a significant reduction in excitability of the somatosensory and anal sphincter motor cortex after chronic SNM treatment (8,20). In-depth investigation of the central neural mechanisms of SNM for FGIDs is essential to improve identification of the most effective stimulation parameters and to determine which patients are the best candidates for different available SNM treatment options including temporary and permanent implants, TENS therapy or combinatory treatments (4,8,11,15,22).
Review of the literature shows that neuromodulation for pain management is more comprehensively researched compared to non-painful conditions. Since SNM initially emerged to treat urinary tract disorders (6,10), FGIDs have received even less attention among the non-painful conditions. However, without fuller understanding of the neurophysiology of SNM, this treatment remains empirical for FGIDs (4). The evolving body of literature on central and peripheral mechanisms of SNM point to the afferent sensory nerves to be the target fibres within the nerve root (10,16). This is currently the most convincing hypothesis of SNM mechanism with supporting evidence from a wider range of studies (4). Efficacy of SNM at voltages at the sensory but sub-motor threshold (4) and clear changes over the somatosensory cortex following SNM administration in both humans and animal models (3,4,8,9,10,11,15) add to the credibility of this hypothesis. Nevertheless, subsensory stimulation as low as 50% of the sensory threshold can still be as effective as stimulation at the sensory threshold (6).
Furthermore, at present it is not yet fully understood why in some studies patients manifest increased cortical excitability after SNM treatment (13, 15) while the opposite effect is observed in others (9, 15, 22). One of the reasons for such disparate results might be due to uncertainty about the optimal sensory response to SNM which makes it difficult to reliably compare data from different groups of researchers (4). This area of SNM research requires serious investigation as it can shed light on not only the central pathways activated by SNM but also on the optimal sensory responses. On the other hand, the conflicting data regarding cortex excitability can be explained by time effect. Reduction in excitability of cortico-anal and somatosensory cortex are reported after chronic SNM treatment while increase in excitability is observed after acute SNM therapy (15).
Although SNM seems to rely on different neural mechanisms for various disorders, there are similarities in the way SNM modulates the underlying neural pathways. The group of nerve fibres that SNM targets and the regions of the brain that it activates resemble each other in disparate conditions. Furthermore, the spinal and supraspinal neural pathways for defecation considerably overlap with those of lower urinary tract (10). This might mean that SNM follows one general neural mechanism that regulates sensory responses depending on the optimal response required to achieve overall homeostasis.
It is known that afferent nerves are critical for sending signals of sensation, fullness or discomfort to the brain to initiate a "response reflex". Such response reflex could be the micturition, the defecation or the withdrawal reflex. The main afferents involved in both micturition and defecation reflexes connect with interneurons in the sacral spinal cord (16). The interneurons in turn synapse with efferent parasympathetic neurons to form spinal reflexes. However, spinal reflexes are modulated by several centres in the brain (10,15,16).
Therefore, it is plausible that SNM modulates response reflexes of any kind by altering the afferent signalling depending on the status quo activity of the afferent nerve fibers innervating the target organ. If any of the response reflexes are excessively active, SNM decreases the afferent signalling to inhibit the reflex and if the reflex is hypoactive, SNM increases the activity of afferent fibres to enhance afferent signalling. This hypothesis could explain the contradictory results obtained from different cortical studies while being consistent with how TENS for pain management modulates the transmission of signals evoked by noxious stimuli. Further research is required to test this proposition.
Acknowledgement: I'd like to thank my supervisor Dr. Jan Huizinga and Ontario Graduate Scholarship.
Harvard Medical Student Review Issue 5 | January 2020